Electric Charge
One of the fundamental forces of nature is
electromagnetism. Electricity and magnetism are both caused by this force. The electromagnetic force affects objects that have
electric charge, and the study of charged objects that are at rest is called
electrostatics.
There are two kinds of electric charge. Benjamin Franklin (1706-1790) was the first to call these kinds of charge
positive and
negative. Two negative charges or two positive charges repel each other, while a positive charge and a negative charge attract each other. This repulsion or attraction is a force that acts between all charged objects.
Electrostatics can be demonstrated using rods and materials of different types. Some common experiments are:
1. Begin with a plastic rod. Rub the rod with fur (real or fake). The rod acquires a negative charge on its surface, while the fur acquires a positive charge. Separate the rod and fur. As the fur is brought close to the rod again, it will attract it.
2. Begin with a glass rod. Rub the rod with silk. The rod acquires a positive charge on its surface, while the silk acquires a negative charge. Separate the rod and silk. As the silk is brought close to the rod again, it will attract it.
3. Begin with two plastic rods. Rub both rods with fur (real or fake). Both rods acquire a negative charge on their surfaces. As the two charged rods are brought close to each other, they repel.
4. Begin with two glass rods. Rub both rods with silk. Both rods acquire a positive charge on their surfaces. As the two charged rods are brought close to each other, they repel.
5. Begin with a plastic rod and a glass rod. Rub the plastic rod with fur (real or fake). Rub the glass rod with silk. As the plastic rod and the glass rod are brought close to each other, they attract.
The Structure of Matter
Charge is a fundamental property of matter, and so understanding where it comes from requires understanding the structure of matter. The atoms that compose all ordinary matter are made of
protons, neutrons, and
electrons. Protons have a positive charge, neutrons have no charge (they are neutral), and electrons have a negative charge. The negative charge of an electron has exactly the same magnitude as the positive charge of a proton. Even though the magnitude of the charges balance, an electron has much less mass than either a proton or neutron. The masses of the particles are,
Mass of electron =
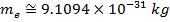
Mass of proton =
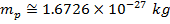
Mass of neutron =
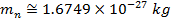
The masses of protons are neutrons are very similar, though the mass of an electron is roughly 2000 times less than either a proton or neutron.
In an atom, protons and neutrons bind together in a small, dense core, called the
nucleus of the atom. The electrons surround the nucleus, though at a larger distance. The nucleus of an atom is on the order of
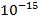
meters in size, but the electrons extend to distances on the order of
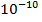
meters.
The number of protons in an atom is called the
atomic number. The atomic number determines what type of element the atom is. For example, all hydrogen atoms have one proton, all carbon atoms have 6 protons, and all gold atoms have 79 protons. The periodic table of the elements is organized by atomic number. The
atomic mass is the combined number of protons and neutrons in the nucleus of the atom.
The charge of an atom is determined by the number of protons and electrons in it. Since neutrons are neutral, they do not contribute to the charge. Most atoms in nature are neutral, as a result of having equal numbers of protons and electrons. If the number of protons and electrons are not equal, the atom is called an
ion. The process of turning a neutral atom into an ion is called
ionization. If one or more electrons is removed, the result is a
positive ion. If an atom gains one or more electrons, it becomes a
negative ion.
The
principle of conservation of charge states that charge cannot be created or destroyed. All neutral atoms are the result of a balance of charges. Ionizing an atom requires adding or removing electrons, but that means they must be removed or added to somewhere else. For example, when a glass rod is rubbed with silk, electrons from the glass rod move from the rod to the piece of silk. The surface of the glass becomes positively charged and the silk becomes negatively charged because electrons moved from one object to the other.
The magnitude of charge of the proton or electron is the smallest quantity of charge found in nature. Every measurable amount of charge is an integer multiple of this number.
Conductors and Insulators
Materials that permit electric charge to move easily within them are called
conductors. In a conductor, atoms can be ionized by removing electrons. The electrons can then move freely throughout the material. The motion of the electrons can carry charge, while the positive ions remain fixed in the material.
Other materials called
insulators do not permit charge to move easily. In an insulator, it is much more difficult to cause ionization, and so there are very few free electrons. Since there are almost no free electrons in the material, insulators do not carry charge.
Most metals are good conductors, and most non-metals are insulators.
Induction
Another way in which charge can move through materials is called
induction. When a charged object is brought close to a neutral conductor, free electrons within the conductor can move, creating a positive charge on one side of the conductor, and a negative charge on the other. These charged regions of the conductor are called
induced charges.
For example, imagine a neutral metal ball on an insulating stand. If a plastic rod with a negative charge is brought close to the ball, free electrons in the ball will be repelled, and will move to the opposite side of the ball. Positive charge will build up on the side of the ball closest to the negatively charged rod.
As another step, imagine a conducting wire is used to connect the negative induced charges on the ball to the ground. This is known as
grounding a charge. The negative charges migrate through the wire to the Earth, which acquires a very slight additional negative charge. Now, the ball has lost free electrons, but the positive induced charge near the rod remains. If the rod is removed, the electrons in the metal will rearrange to evenly distribute the positive charge. In this way, the ball can acquire a net positive charge.
Induction can also affect insulators, though not to the same degree. A charged object brought close to an insulator can cause a shift of charge in the molecules of the insulator. This is called
polarization. The polarization occurs whether the charged object is positive or negative. The result is that an attractive force can be exerted on an insulator by either a negatively or positively charged object. This is the reason an object with a small surface charge like a balloon can attract small pieces of paper, for example.
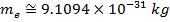
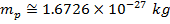
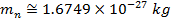
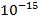 meters in size, but the electrons extend to distances on the order of
meters in size, but the electrons extend to distances on the order of 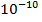 meters.
meters.