Aluminum Sulfide Formula
Aluminum sulfide, also known as dialuminium trisulfide, is an inorganic compound used as raw material in the synthesis of the largely-used hydrogen sulfide.
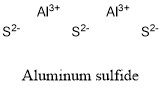
Formula and structure: Aluminum sulfide chemical formula is Al2S3 and the molar mass is 150.16 g mol-1. Sometimes this sulfide can be found in its hydrate form, with a variable number of water molecules hydrating it. The compound is formed by two cation Al3+ and three anions S2-. Aluminum sulfide has several crystal structures, some of them are the tetragonal, hexagonal and trigonal. Its chemical structure can be written as below, in the common representations used for organic molecules.
Occurrence: Aluminum sulfide is not found in nature.
Preparation: Aluminum sulfide is prepared by the reaction of the aluminum metal with sulfur powder. They are heated to ignition to form the sulfide though a highly exothermic reaction:
2 Al + 3 S → Al2S3
Physical properties: Aluminum sulfide is a gray to black solid. Its density is 2.02 g mL-1. The melting point of this compound is 1100 °C and the boiling point is 1500 °C, at this temperature it sublimes. Aluminum sulfide decomposes in water and is insoluble in acetone.
Chemical properties: Aluminum sulfide, similar to other metal sulfides, is slightly soluble in water and largely soluble in acid solutions. In presence of sodium or potassium bases, it can react to form sodium sulfide, which is an irritant and can cause irritation in skin and eyes.
Uses: Aluminum sulfide is used in the preparation of hydrogen sulfide, a compound that is largely used in the chemical industry. Moreover, aluminum sulfide is used in the manufacturing of cathodes that contain lithium-sulfur solid-state batteries. Along with these applications, aluminum sulfide is also part of the production processes of compounds as ethanethiol.
Health effects / safety hazards: Aluminum sulfide is highly toxic and dangerous for the health. It is poisonous by inhalation. It can cause severe damage in the nervous system and the accumulation in the body can also cause blood damage.
|
Related Links: |
