Cadmium (II) Fluoride Formula
Cadmiun (II) fluoride or cadmium difluoride is an inorganic salt which is an important source of cadmium and it is also used in the pharmaceutical industry.
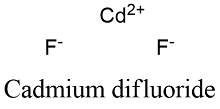
Formula and structure: The cadmium fluoride chemical formula is CdF2 and its molar mass is 150.410 g mol-1. Cadmium fluoride molecule is formed by the cadmium cation Mg+2 and the fluoride anion F-. The cadmium fluoride lattice is isometric cubic, similar to the mineral fluorite (CaF2). Its chemical structure can be written as below, in the common representations used for organic molecules.
Occurrence: Cadmium fluoride can be found in nature, in some geological formations. However, it is mostly prepared through chemical synthesis.
Preparation: Cadmium fluoride is prepared from several methods; the most used in chemical industry are: the reaction of fluoride in gas or hydrogen fluoride and a salt or metal of cadmium:
CdO + 2 HF → CdF2 + H2O
It can also be prepared from the dissolution of cadmium carbonate in solution of HF or form the reaction between the cadmium chloride and ammonium fluoride.
Physical properties: Cadmium fluoride is grey to white, odorless crystalline solid. Its density is 6.33 g mL-1. The melting point is 1110 ºC and its boiling point is 1749 ºC. It is poorly soluble in water, soluble in acid and insoluble in ethanol and ammonia.
Chemical properties : Cadmium fluoride is formed by the cation Cd`2+ that is part of the element known as metalloids; these have properties between the metal (as gold, iron, copper, etc) and the non-metals (as carbon, sulfur, etc). The metalloids are semi-conductor due their properties between the two groups. Remarkably, the cadmium compounds are being studying due its capacity as semiconductor and to apply in nanotechnology.
Uses: Cadmium fluoride is used in anoparticles synthesis and research owing its capacity as semiconductor. Moreover, when combining with other elements such as the earth elements, it can b transformed in an electronic conductor. It is also used in organic chemistry to perform synthesis.
Health effects / safety hazards: Cadmium fluoride is extremely toxic if swallowed or inhalet. It can be fatal when is in our body. It can also cause genetic defects and cancer and damage the fertility. It is also very toxic to aquatic and soil life. It is not flammable.
|
Related Links: |
