Calcium oxide Formula
Calcium oxide, also known as quicklime, is a substance largely used in chemical industry as intermediary in the cement and concrete production and also in the soda ash synthesis process.
Formula and structure: Calcium oxide chemical formula is CaO and its molar mass in 56.0774 g mol-1. The molecule is formed by the calcium cation Ca+2 and the oxygen anion O-2, which form a ionic bond. The calcium oxide ionic lattice is cubic and similar to NaCl lattice, with an ion surrounded by 6 opposite charge-ion. Its chemical structure can be written as below, in the common representations used for organic molecules.
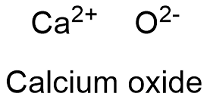
Occurrence: Calcium oxide is found in nature as part of mineral Lime, which is a material composed by calcium, magnesium and aluminium carbonates, oxides and hydroxides.
Preparation: Calcium oxide is mostly obtained from the lime through heating until the decomposition of material into CaO and CO2:
CaCO3(s) → CaO(s) + CO2(s)
Physical properties: Calcium oxide is an odorless, white to yellow, gray or brown granular powder. Its density is 3.34 g mL-1. The melting and boiling points are 2613 ºC and 3850 ºC, respectively. It is soluble in water, reacting to form calcium hydroxide. Calcium oxide is insoluble in methanol and ethyl ether.
Chemical properties: Calcium oxide has a vast chemical reactivity. It can react in many different ways, two examples are:
Calcium oxide is used to obtain building material because when it is mixture with water can reacts to form calcium hydroxide, which rapidily forms a harden material:
CaO + H2O → Ca(OH)2
It can also react with fossil sources to produces calcium carbide:
2 CaO(s) + 5 C(s) → 2 CaC2 (s) + CO2 (g)
Uses: Calcium oxide is very used in the production of building materials as cement and concrete, similar to other calcium compounds as calcium sulfate. Moreover, calcium oxide is used in the manufacturing and production of substances as soda ash, bleaching liquids, dry and desiccants and calcium compounds. It is also used to produce fertilizers and as additive in the water treatment/purification.
Health effects / safety hazards: Calcium oxide can cause severe eye and mucous irritantion. It is extremely toxic by ingestion and is corrosive to the skin. When large quantities of calcium oxide are dissolved in water, the resulting reaction is highly exothermic. It is corrosive and can react violently with ethanol, phosphorus pentoxide and acids.
|
Related Links: |
