Ferric sulfate Formula
Ferric sulfate, also known as iron (III) sulfate, is an inorganic salt used on water purification for industrial wastes and also as oxidizing agent.
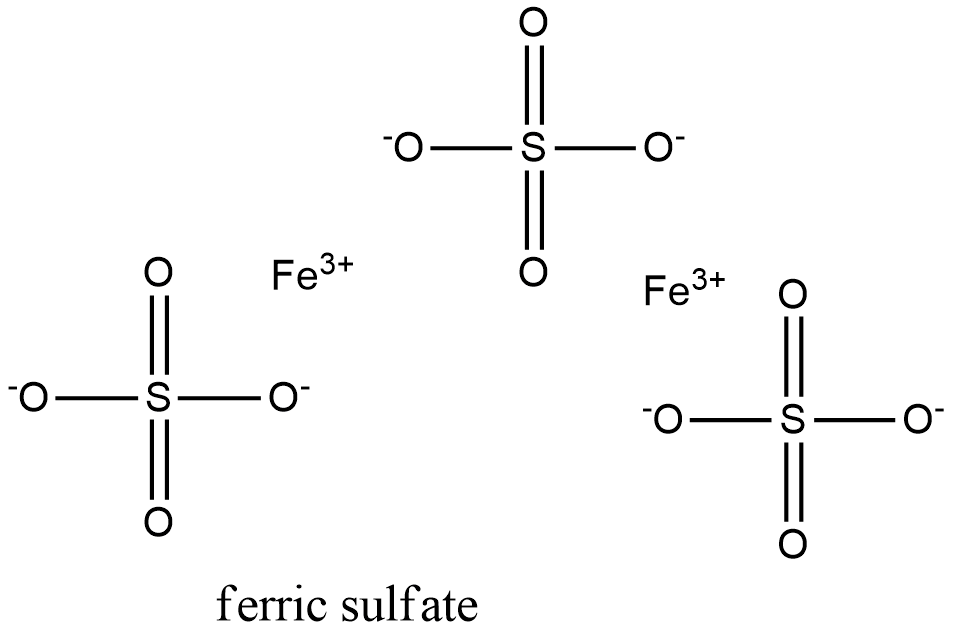
Formula and structure: The ferric sulfate chemical formula is Fe2(SO4)3. Its molecular formula is Fe2O12S3 and its molar mass is 399.858 g mol-1. The molecule is formed by the cation Fe+3 and the sulfate anion SO4-2. It is frequently found in nature as hydrated salt and the most common are the penta- and nonahydrated salts, which has the molar mass of 489.960 g mol-1 and 562.000 g mol-1, respectively. The molecules of ferric sulfates form rhombic or rhombohedral crystals. Its chemical structure can be written as below, in the common representations used for organic molecules.
Occurrence: Ferric sulfate is found in a wide variety of minerals. It is mainly found in the minerals pyrite and marcasite, where the ferric sulfate is connected with Fe0. Other minerals as coquimbite, kornelite and lausenite are sources of nona-, hepta- and pentahydrated ferric sulfate. Moreover, ferric sulfate may be found minerals such as Mikasite, where it forms a mixture with aluminium.
Preparation: Ferric sulfate is mostly extrated from nature, however it is also prepared by treating ferrous sulfate and an oxidizing agent with sulfuric acid at elevated temperatures (I). It can also be prepared by evaporating ferric oxide and sulfuric acid.
2FeSO4 + H2SO4 + H2O2 → Fe2(SO4)3 + 2H2O (I)
Ferric sulfate solutions are prepared by injecting chlorine gas in a ferrous sulfate solution.
Physical properties: Ferric sulfate is a white to gray powder (anhydrous salt) or a yellow crystalline solid (hydrated salt). Ferric sulfate odor can be varied accord to the number of water molecules present in the salt. Its density is 3.097 g mL-1 (anhydrous) and its melting point is 480 ºC (anhydrous) and 175 ºC (nonahydrate). It is slowly soluble in water. It is insoluble in sulfuric acid and ammonia and also in acetone and ethyl acetate.
Chemical properties: Ferric sulfate can be used in water purification due the trivalent iron Fe+3 is a cation that helps to minimized or preventing the formation of hydrogen sulfide (H2S). This cation also acts as a coagulant in phosphorous removal. The Fe+3 is also responsible by the capacity of ferric sulfate to oxide other compounds because it can lose 1 electron to form Fe+2.
Uses: Ferric sulfate is used in the industry in the water and wastewater treatment operations due its capacity as flocculant and coagulant and to eliminate odor from sulfur compounds. Ferric sulfate is used as solids separation agent and oxidizing agent. Moreover, this salt is used in pigment industry. In medicine, it can be used as astringent and styptic.
Health effects/ Hazard safety: Ferric sulfate may cause eyes, throat and nose irritation by inhalation and mouth and stomach by ingestion. It is not flammable, however, when heated, ferric sulfate emits toxic fumes of iron and sulfur oxides.
|
Related Links: |
Related Topics
Normality Formula
