Octanoic acid Formula
Octanoic acid, most commonly known as caprylic acid (named from the Latin capra that means goat) is a natural carboxylic acid found in goat milk and coconuts oil.
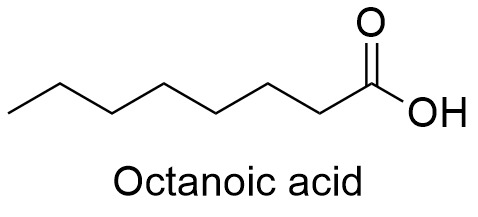
Formula and structure: The chemical formula of Octanoic acid is (C8H16O2; the structure is a eight-carbon linear chain without any branch, in the carbon 1, there is an carboxylic acid group, having the final structure CH3(CH2)6COOH. It molecular mass is 144.21 g mol-1. The chemical structures can be written as below, in the common representations used for organic molecules.
Occurrence: Octanoic acid is found in mammals' milks, coconuts and palm oils.
Preparation: Octanoic acid can be found as naturally occurring in nature; however several methodologies have been developed in order to increase its production. When extracted from coconut oil, the oil is reacted through a saponification and after that, a distillation to recovery the pure Octanoic acid. Chemically, it can be prepared from 1-heptene, which is oxidized to octanol and then to acid.
Physical properties: Octanoic acid is a colorless, oily with fruity-acid smell liquid. Its melting point is 16.7°C and its boiling point is 239.7°C. The density is 0.910 g mL-1. It is not soluble in water, but it is highly soluble in ethanol, methanol, ether, chloroform and other organic solvents.
Chemical properties: Octanoic acid is classified as a fatty acid, which is a group of acid naturally find in a wide number of natural products. These acids have a structure formed by 1 carboxylic acid group to which is attached a long carbon backbone. The unsaturated chain is the responsible for the low solubility exhibited by the fatty acids in water.
Uses: Octanoic acid is used as a raw material to obtain esters. It is a natural component of milks of different mammals and some vegetable oils. It is also used as pesticide and antimicrobial agent and widely used in personal care manufacturing. Moreover, it is a disinfectant in hospitals and industrial facilities. It has been also used as a supplement for some athletes.
Health effects/safety hazards: Octanoic acid can be irritant in large quantities. However, in small quantity it is safe for human and animal consume. It is highly corrosive to metals and tissue. It can burn but not easily.
|
Related Links: |
