Perchloric Acid Formula
Perchloric acid is an inorganic acid usually prepared as an aqueous solution that is used to prepare perchlorate salts that are later used in chemical industry.
Formula and structure: The perchloric acid chemical formula is HClO4 and the molar mass is 100.46 g mol-1. The acid structure is formed by a chlorine atom in the center to which 3 oxygen atoms are bound through double bonds, a fourth bond is formed between the chlorine and a hydroxyl. This hydroxyl has the acidic proton which the acid characteristic to the molecule. Its chemical structure can be written as below, in the common representations used for organic molecules.
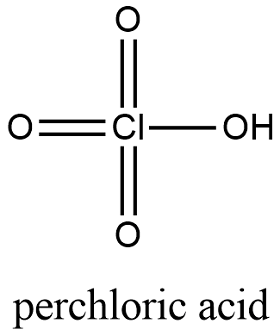
Occurrence: Perchloric acid cannot be found in nature. It is prepared as described below.
Preparation: perchloric acid can be prepared from potassium perchlorate and sulfuric acid or through the reaction of sodium perchlorate and HCl at room temperature:
NaClO4 + HCl → NaCl + HClO4
Another reaction is the electrolytic oxidation of HCl with the subsequent irradiation of chlorine dioxide.
Physical properties: Perchloric acid uses to be a colorless and odorless 70% solution of water. It is miscible in water and its density is 1.768 g mL-1. Its melting and boiling point are -17 °C and 203 °C respectively. It can form azeotrope with water.
Chemical properties: Perchloric acid is a strong acid, with a pKa of -10, it means, the conjugated base of the Perchloric acid is stable and it is almost completely dissociated in aqueous solutions:
HClO4 + H2O → H3O+ + ClO4-
Thus, it is widely used in reaction where a strong acid is required.
Uses: Perchloric acid is used to prepare perchlorate salts such as ammonia perchlorate which is used as a rocket fuel. It is a strong acid that is used to get an acid aqueous solution or medium in chemical reactions in laboratory or industrial scale.
Health effects / safety hazards: Perchloric acid can cause fire or explosion. It is a strong oxidizer and it is extremely corrosive to metals. It is harmful if swallowed and cause eyes and skin damage.
|
Related Links: |
