Salinomycin Formula
Salinomycin is an antibiotic used on the veterinary treatment of infections. Salinomycin is an organic compound which is also a potential antitumor agent.
Formula and structure: The salinomycin chemical formula is C42H70O11 and its molar mass is 751.011 g mol-1. The molecule is a polyether ionophore, which is formed by 4 pyran rings and 1 furan ring and 17 chiral centers. Its chemical structure can be written as below, in the common representations used for organic molecules.
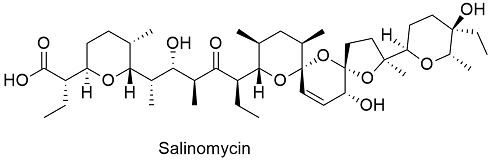
Occurrence: Salinomycin is naturally produced by the genus of bacteria Streptomyces, particularly Streptomyces albus.
Preparation: There are few methods to produce salinomycin through synthetic methods. It has been produced during many years almost exclusively by bacterial fermentation. The main difficult to produce by synthesis is the number of sterocenters of the molecule, which represent an enormous challenge for the chemists.
Physical properties: Salinomycin is a white or light yellow crystalline solid. Its density is 1.2 g mL-1 and its melting point is 140 ºC and at higher temperatures, it decomposes. Salinomycin is soluble in ethanol methanol, diethyl ether, ethyl acetate and bezene. It is poorly soluble in water.
Chemical properties: Salinomycin structure can act as antibiotic due to the multiple ether moieties, which form negative density that attracts cations such as potassium and sodium from the cellular cytoplasm. Moreover, the exterior of the salinomycin molecule forms a hydrophobic coat which allows the passage of the cations to the exterior of the cells and leading the cellular death by losing of cations.
Uses: Salinomycin is used as anticoccidial drug to cattle and other species. It is not used in human because is toxic and instead is used other antibiotics as lasalocid or nigericin. Since 2009, many studies have been developed for many researchers to evaluate the application of salinomycin in the treatment of some type of cancers such as the ovarian cancer. Additionally, salinomycin is used as standard in some analytical determination through the mass spectroscopy technique.
Health effects / safety hazards: Salinomycin is toxic when consumed in large quantities and can cause the death. It is not flammable. The antibiotic can decompose at high temperatures.
|
Related Links: |
