Silver Sulfide Formula
Silver sulfide, also known as disilver sulfide, is an inorganic salt used as a photosensitizer in photography.
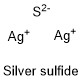
Formula and structure: The silver sulfide chemical formula is Ag2S. The molar mass is 247.80 g/mol. The molecule is formed by two cation silver Ag+ and one sulfide anion S2-. The two cations are bound to the sulfide anion through an ionic bond. The molecule can have different geometry: face-centered-cubic (> 586 °C), monoclinic (<179 °C) and body centered cubic (> 180 °C). Its chemical structure can be written as below, in the common representations used for organic molecules.
Occurrence: Silver sulfide is found as forming a thin layer of corrosion in silver(tarnish).
Preparation: Silver sulfide can be prepared from metallic silver, to which a stream of hydrogen sulfide gas is pass through.
2Ag + H2S → Ag2S + H2
Physical properties: Silver sulfide is a gray to black odorless crystal. Its density is 7.23 g/mL at room temperature. Its melting point is 836 °C and above this temperature, it decomposes. Silver sulfide is soluble in some strong acids as HCN and nitric acids. In other acids (mostly weak acids) and alkalies is insoluble.
Chemical properties: Silver sulfide is a good electrical conductor. Many silver salts and the silver metallic are consider the best conductors, this Ag2S can be used in some electrical applications. Even when the salt is a solid with a high melting point, it is slower than other salts. The cause for it comes from the fact that the ionic character of the bond Ag-S is not enough strong, with a difference of electronegativity of less than 10%.
Uses: Silver sulfide can be used as agent for revealing photos. It is also used as a tarnish to protect silver from corrosion. It is used in chemical determination of nitrate ion in solution, where some drops of solution of silver sulfide are added and then, silver nitrate precipitates:
2NaNO3 + Ag2S → 2AgNO3(solid) + Na2S
Health effects / safety hazards: Silver sulfide is not flammable and does not react with other chemical compounds. It is not considered harmful to health.
|
Related Links: |
