Sodium chloride Formula - Sodium chloride Uses, Properties, Structure and Formula
Sodium chloride is one of the most well-known and widely used chemicals, also known as table salt.
Formula and structure: The chemical formula of sodium chloride is NaCl and its molar mass is 58.44 g/mol. It is an ionic compound consisting of a sodium cation (Na+) and a chloride anion (Cl-). Solid NaCl has a crystalline structure, in which each Na+ ion is surrounded by six chloride ions in an octahedral geometry.
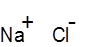
Occurrence: Sodium chloride is present in the sea and ocean waters, giving them their saltiness. About 1-5% of seawater is made of sodium chloride. It is also found as the mineral halite or rock salt.
Preparation: Salt is produced on large scales by evaporation of sea water or the salt water (brine) from salt lakes and brine wells. As seawater contains several other salts (of calcium, magnesium and other elements), the evaporation process is carried out carefully so that the different salts precipitate out at different times based on their solubility. Another major production method is by mining the rock salt reserves.
Physical properties: Sodium chloride is a white crystalline solid with a density of 2.16 g/mL, and a melting point of 801 °C. It is also available as aqueous solutions of different concentrations, called saline solutions.
Chemical properties: Sodium chloride is readily soluble in water and other polar solvents. It is a stable solid. It only decomposes at high temperatures to give toxic fumes of hydrochloric acid (HCl) and disodium oxide (Na2O).
Uses: Sodium chloride is best known as table salt and is used widely in the food industry for flavoring and preservation. It is also used in the production of many important chemicals, including sodium hydroxide, sodium carbonate, baking soda, hydrochloric acid, etc. It also has applications in oil refineries, textiles industry, paper and pulp industry, fire-retardants, rubber industry and in road constructions. Another important use is the de-icing of roads and sidewalks in cold and snowy regions. Saline solutions are used for many medical purposes too.
Health effects/safety hazards: At low concentrations, sodium chloride is non-toxic and non-hazardous, and is an important electrolyte source for the body. High consumption of salt over the long term can cause imbalance of electrolytes in the body. Swallowing high concentrations of it can cause vomiting, nausea, diarrhea and dehydration. It can also irritate the eyes and cause eye damage at high concentrations.
|
Related Links: |
