Valinomycin Formula
Valinomycin, also known as Cyclo (L-Val-D-HyIva-D-Val-L-Lac-)3, is a cyclododecadepsipeptide ionophore antibiotic produced by bacteria and used as treatment of respiratory-syndrome coronavirus and it is also used in biochemical studies.
Formula and structure: Valinomycin is a large molecule and the chemical formula is C54H90N6O18; its molar mass is 1111.32 g mol -1. Valinomycin is a large cycle formed by some sub-units such as: D- and L-valine, D-alpha-hydroxyisovaleric acid, and L-lactic acid which are bounded formed a 36-members ring. Its chemical structure can be written as below, in the common representations used for organic molecules.
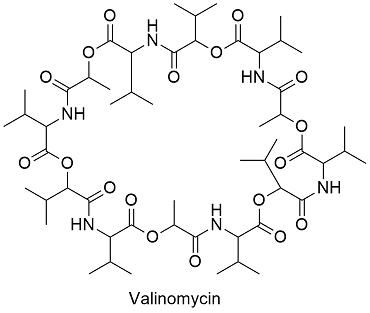
Occurrence: Valinomycin is biosynthesized by bacteria such as Streptomyces fulvissimus. These bacteria produced the antibiotic as a defense mechanism against other bacteria or fungi.
Preparation: Valinomycin is only produced by bacteria of the genus Streptomyces and it is not found in other organisms in nature. Due to the complex structure of valinomycin it is not produced by chemical methods.
Physical properties: Valinomycin is a white shinny crystalline solid. Its density is 1.06 g mL -1. Valinomycin melting point is 190 ºC and above this temperature it decomposes. It is insoluble in water but is soluble in ethanol, ether, benzene, chloroform, acetic acid and acetone.
Chemical properties: Valinomycin is a macrocyclic molecule that can act as antibiotic due to induce the cells death through a depolarization of the cell. The depolarization is caused because the multiple carbonyl groups that form the core of the cycle attract potassium and sodium cations and transport them outside the cells. The exterior of the molecule of valinomycin is a hydrophobic coat which allows the passage across the cellular walls.
Uses: Valinomycin is an antibiotic used to induce cell death through K + selective transport. Additionally valinomycin is used as a standard compound to study many biochemical phenomena, for example, experiments of membrane vesicles.
Health effects / safety hazards: Valinomycin is a neurotoxine, it is extremely toxic and fatal if swallowed in large quantities. It can also be absorbed by the skin and in this case, it is also fatal. It is not flammable but when heated to decomposition, it emits toxic fumes of nitrogen oxides.
|
Related Links: |
